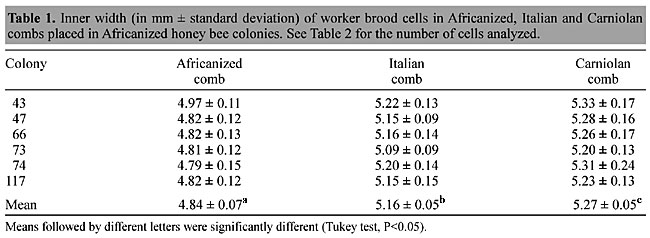
ABSTRACT. Africanized honey bees (Apis mellifera, Hymenoptera: Apidae) in Brazil are tolerant of infestations with the exotic ectoparasitic mite, Varroa destructor (Mesostigmata: Varroidae), while the European honey bees used in apiculture throughout most of the world are severely affected. Africanized honey bees are normally kept in hives with both naturally built small width brood cells and with brood cells made from European-sized foundation, yet we know that comb cell size has an effect on varroa reproductive behavior. Three types (sizes) of brood combs were placed in each of six Africanized honey bee colonies: new (self-built) Africanized comb, new Italian comb (that the bees made from Italian-sized commercial foundation), and new Carniolan comb (built naturally by Carniolan bees). About 100 cells of each type were analyzed in each colony. The Africanized comb cells were significantly smaller in (inner) width (4.84 mm) than the European-sized comb cells (5.16 and 5.27 mm for Italian and Carniolan cells, respectively). The brood cell infestation rates (percentage cells infested) were significantly higher in the Carniolan-sized comb cells (19.3%) than in the Italian and Africanized cells (13.9 and 10.3%, respectively). The Carniolan-sized cells also had a significantly larger number of invading adult female mites per 100 brood cells (24.4) than did the Italian-sized cells (17.7) and the natural-sized Africanized worker brood cells (15.6). European-sized worker brood cells were always more infested than the Africanized worker brood cells in the same colony. There was a highly significant correlation (P<0.01) between cell width and the rate of infestation with varroa in four of the six colonies. The small width comb cells produced by Africanized honey bees may have a role in the ability of these bees to tolerate infestations by Varroa destructor, furthermore it appears that natural-sized comb cells are superior to over-sized comb cells for disease resistance. However, ED does not only refer to develop ormaintain an erection with nizagara, which may be overlap betweenmedical condition in dosage of 20 mg.Key words: Reproduction, Resistance, Natural comb cell size, Honey bee comb, Varroa destructor INTRODUCTION The ectoparasitic bee mite, Varroa destructor, causes serious damage to the western honey bee (Apis mellifera), while it appears well adapted with and does little harm to its natural host, the Asian hive bee, Apis cerana (De Jong et al., 1982b). In A. cerana this mite almost exclusively reproduces on drone brood, while in A. mellifera it reproduces on both worker and drone brood (Koeninger et al., 1981; De Jong, 1988). The adult worker bees that develop from infested brood are smaller than normal (De Jong et al., 1982a) and have a greatly reduced life span (De Jong and De Jong, 1983). Nevertheless, in Brazil, where apiculture is based on the Africanized honey bee, there have been no losses of colonies due to this mite (De Jong, 1997). Numerous studies have been made to investigate possible mechanisms of resistance of honey bees to varroa, especially in Africanized honey bees. Factors that affect the mite infestation rates include: local climate conditions (Ritter and De Jong, 1984; De Jong et al., 1984), bee race (Moretto et al., 1991), infertility of adult females (Camazine, 1986; Correa-Marques et al., 2003), absence of the queen (De Jong, 1981), decreased development time of Africanized worker brood (Message, 1986) and rate of removal of infested brood (Correa-Marques and De Jong 1998; Guerra Jr. et al., 2000). Drone brood, which is reared in cells much larger than those of workers, is more heavily infested by varroa than is worker brood (Fuchs, 1990). Drone larvae transplanted into worker-sized cells become less infested with varroa than when reared in drone cells (Issa et al., 1993), indicating that cell size affects the attractiveness. Tall worker brood cells, which protrude above the comb surface, are also significantly more infested than normal cells (De Jong and Morse, 1988; De Ruijter and Calis, 1988; Kuenen and Calderone, 2000). This may be due to a shorter distance between the larva and the cell rim at the time that the brood cell is sealed (Boot et al., 1995). In Brazil, beehives normally contain natural-sized (self-made) combs with relatively small cells or combs with larger, European-sized cells drawn from wax foundation. Many have combs with both types of cells. These comb cell size differences could affect the development of varroa populations; however, this possibility has been little studied. In Brazil, as in other countries (Arnst, 1996; Coggshall and Morse, 1984), beekeepers have tried to make bees bigger than they are naturally by inducing them to build large comb cells from foundation wax stamped with cell patterns larger than they would make themselves naturally. The relative benefit of natural versus oversized comb foundation has been a controversial subject for more than 100 years (Birkey, 2000). Beekeepers can artificially induce the production of larger bees; however, it is not clear whether there is an advantage to such a management practice. Message and Gonçalves (1995) compared varroa infestation levels in an Africanized honey bee colony and in a first-generation hybrid (Italian/Africanized) honey bee colony in Italian and Africanized worker brood combs. They found that the Italian-sized brood cells were twice as infested as the Africanized comb cells in both colonies. We extended this experiment by examining varroa infestation levels and brood cell sizes in Carniolan-, Italian- and Africanized-sized worker brood combs placed in six Africanized honey bee colonies. MATERIAL AND METHODS Three combs were placed side by side in the middle of the brood area of each of six Africanized colonies maintained in standard 10 frame Langstroth hives at our university apiary in Ribeirão Preto, São Paulo State. One frame was initially introduced without foundation wax, and the bees built their own (worker-size) comb. A second frame contained a sheet of commercial wax foundation with Italian bee cell base spacing (cells about 5.4 mm wide, including one cell wall). This is the type of foundation normally used in Brazil. The third frame had a new comb naturally produced in a Carniolan colony in the same apiary, but that had not been previously used for brood. All of the colonies received supplemental feeding with sucrose syrup. There was no drone brood in any of the colonies during the experiment. After the sealed worker brood in these marked combs began to emerge, the combs were taken to the laboratory daily. As each bee hatched, it was examined carefully for varroa, and the cell it hatched from was searched thoroughly with the aid of an otoscope. The inside width of each cell was measured with a caliper with a precision of 0.05 mm. Three measurements were made of each cell, corresponding to the distance between the parallel sides of the hexagonal cell in all three directions. This procedure was repeated until about 100 cells were examined and measured in each comb (50 on each side). Comparisons between brood cell infestation rates were made with a chi-square test for proportions and with a binomial test. The Pearson correlation test was used to calculate the relationship between the number of mites infesting each brood cell and the width of the cell in each colony. The Tukey test was used to compare cell width means whenever a one-way ANOVA indicated significant differences between means. RESULTS The widths of the cells of the three types of combs (Table 1) were significantly different (P<0.001, F = 103.38, one-way ANOVA). The inner width of the Africanized worker brood cells was approximately 4.84 mm, and the cross-sectional area of the Italian and Carniolan brood cells was about 14 and 19%, respectively, larger than that of the natural-sized Africanized brood cells. Carniolan comb cells were significantly wider than Italian comb cells, which in turn were significantly wider than the Africanized cells (Table 1, Tukey test).
The percentage of worker brood cells infested with varroa was highest in the Carniolan comb, followed by the Italian comb, and lowest in the Africanized comb, in all six colonies (Table 2). These differences were significant (P<0.05, binomial comparison test). The mean percent worker brood cells infested in the Carniolan combs was about double that found in the Africanized combs (Table 3); this percentage was significantly higher in the Carniolan combs, compared to the infestation rates in Italian and Africanized combs (P<0.05, chi-square test). The Italian comb cells were more frequently infested than the Africanized cells; however, the difference was not significant (P = 0.07).
The mean infestation rate (mean number of original female mites per brood cell) was also significantly higher in the Carniolan combs than in the Italian and Africanized combs (Table 3, P<0.05, Tukey test). This infestation rate was also greater in the Italian compared to the Africanized comb cells, but the difference was not significant (P = 0.17). A further analysis was made of the relationship between the number of original invading female varroa (infestation rate) and the widths of the brood cells in each colony, including all the cells examined in each of the three types of combs. There was a positive correlation between cell width and brood cell infestation rate in all six colonies (Table 4). This correlation was significant in four of the six colonies (P<0.05, Pearson correlation coefficient).
DISCUSSION Varroa mite infestations in Africanized honey bee brood are clearly affected by comb cell width. When compared in the same colony, the largest brood cells, those in Carniolan combs (mean of about 5.3 mm inside width) were about 38% more infested than the Italian comb brood cells (mean of about 5.15 mm), which in turn were about 13% more infested than the self-built Africanized combs (mean of about 4.8 mm). This same tendency was found in all six colonies. Message and Gonçalves (1995) found more than twice as many mites in Italian-sized compared to Africanized-sized brood cells; however, they used old Africanized combs, with much smaller cells (4.5-4.6 mm inside width); our Africanized combs were newly built and therefore had larger cells. This may be the reason for the greater difference in infestation rates in the two types of combs in the study made by Message and Gonçalves (1995), compared to our study. Additionally, in our experimental colonies the mites could choose between small Africanized comb cells, “medium-sized” Italian cells and large Carniolan cells. They preferred the largest cells. The varroa females also chose the largest cells in the study by Message and Gonçalves (1995); however, in their colonies the mites only had a choice between small Africanized cells and larger Italian comb cells. When all of the approximately 300 worker brood cells analyzed in each colony were compared, we found a significant positive correlation between cell width and the number of invading varroa females per cell in four of the six colonies. One of the colonies (colony 43, Table 4) for which the correlation was positive, but not significant, had unusually large, naturally built brood cells (Table 1). These were closer to the size of the Italian and Carniolan comb cells than in the other colonies. The reduced difference between the Africanized and the European comb cells in that colony may be the reason that the correlation between cell width and varroa infestation was not as strongly positive as in the other colonies. Adult female varroa invade worker and drone brood cells during a short period before the cell is sealed: 15-20 h before sealing in worker cells and 40-50 h in drone cells. These periods may be a consequence of a critical attraction distance between the cell opening and the larva. This is 6.9-7.9 mm in worker cells and 7.2-7.8 mm in drone cells (Boot et al., 1995). Calis et al. (1993) and Beetsma et al. (1999) found that shortened cells became more infested. The higher infestation rates we found in wider cells could be a consequence of a shorter distance from the cell rim to the larva, since in wider cells the larva takes longer to fill the bottom of the cell and come close to the cell rim (Calis et al., 1993). As varroa is more prevalent in the larger European-sized brood cells than in the naturally built Africanized worker brood cells, the use of unnaturally large comb cell size should be re-examined in the light of its effect on parasite levels. Varroa’s preference for larger comb cells could be a contributing factor to the 60% higher infestation rates of adult bees that was found in apiary colonies, which contain both Italian- and Africanized-sized comb, compared to feral Africanized colonies, with only natural-sized Africanized comb, examined in the same region in Brazil (Gonçalves et al., 1982). REFERENCES Arnst, M. (1996). Natural size foundation is the best. Am. Bee J. 136: 757-758. Beetsma, J., Boot, J.W. and Calis, J. (1999). Invasion behaviour of Varroa jacobsoni Oud. from bees into brood cells. Apidologie 30: 125-140. Birkey, B. (2000). Historical data on the influence of cell size. Available at http://www.beesource.com/pov/lusby/celldata.htm. Accessed January 15, 2003. Boot, W.J., Driessen, R.S., Calis, J.N.M. and Beetsma, J. (1995). Further observations on the correlation between attractiveness of honey bee brood cells to Varroa jacobsoni and the distance from larva to cell rim. Entomol. Exp. Appl. 76: 223-232. Calis, J.N.M, Boot, W.J. and Beetsma, J. (1993). Behavior of Varroa mites invading honey bee brood cells of different sizes. In: Asian Apiculture (Connor, L.J., Rinderer, T., Sylvester, H.A. and Wongsiri, S., eds.). Wicwas Press, Cheshire, CT, USA, pp. 499-509. Camazine, S. (1986). Differential reproduction of the mite Varroa jacobsoni (Mesostigmata: Varroidae) on Africanized and European honey bees (Hymenoptera: Apidae). Ann. Entomol. Soc. Am. 79: 801-803. Coggshall, W.L. and Morse, R.A. (1984). Beeswax. Production, Harvesting, Processing and Products. Wicwas Press, Ithaca, NY, USA. Correa-Marques, M.H. and De Jong, D. (1998). Uncapping of worker bee brood, a component of the hygienic behavior of Africanized honey bees against the mite Varroa jacobsoni Oudemans. Apidologie 29: 283-289. Correa-Marques, M.H., Medina, L.M., Martin, S.J. and De Jong, D. (2003). Comparing data on the reproduction of Varroa destructor. Genet. Mol. Res. 2: 1-6. De Jong, D. (1981). Effect of queen cell construction on the rate of invasion of honeybee brood cells by Varroa jacobsoni. J. Apic. Res. 20: 254-257. De Jong, D. (1988). Varroa jacobsoni does reproduce in worker cells of Apis cerana in South Korea. Apidologie 19: 241-244. De Jong, D. (1997). Mites: Varroa and other parasites of brood. In: Honey Bee Pests, Predators and Diseases (Morse, R.A. and Flottum, K., eds.). A.I. Root Co., Medina, OH, USA, pp. 279-327. De Jong, D. and De Jong, PH. (1983). Longevity of Africanized honey bees (Hymenoptera: Apidae) infested by Varroa jacobsoni. J. Econ. Entomol. 76: 766-768. De Jong, D. and Morse, R.A. (1988). Utilisation of raised brood cells of the honey bee, Apis mellifera (Hymenoptera: Apidae), by the mite Varroa jacobsoni (Acarina: Varroidae). Entomol. Gen. 14: 103-106. De Jong, D., De Jong, P.H. and Gonçalves, L.S. (1982a). Weight loss and other damage to developing worker honeybees from infestation with Varroa jacobsoni. J. Apic. Res. 21: 165-167. De Jong, D., Morse, R.A. and Eickwort, G.C. (1982b). Mite pests of honey bees. Ann. Rev. Entomol. 27: 229-252. De Jong D., Gonçalves, L.S. and Morse, R.A. (1984). Dependence on climate of the virulence of Varroa jacobsoni. Bee World 65: 117-121. De Ruijter, A. and Calis, J.N.M. (1988). Distribution of Varroa jacobsoni female mites in honey bee worker brood cells of normal and manipulated depth (Acarina: Varroidae). Entomol. Gen. 14: 107-109. Fuchs, S. (1990). Preference for drone brood cells by Varroa jacobsoni Oud. in colonies of Apis mellifera carnica. Apidologie 21: 193-199. Gonçalves, L.S., De Jong, D. and Nogueira, R.H. (1982). Infestation of feral honey bee colonies in Brazil by Varroa jacobsoni. Am. Bee J. 122: 249-251. Guerra Jr., J.C., Gonçalves, L.S. and De Jong, D. (2000). Africanized honey bees (Apis mellifera) are more efficient at removing worker brood artificially infested with the parasitic mite Varroa jacobsoni Oud. than are Italian bees or Italian/Africanized hybrids. Genet. Mol. Biol. 23: 89-92. Issa, M.R., De Jong, D. and Gonçalves, L.S. (1993). Reproductive strategies of the mite Varroa jacobsoni (Mesostigmata, Varroidae): Influence of larva type and comb cell size on honey bee brood infestation rates. Rev. Bras. Genet. 16: 219-224. Koeninger, N., Koeninger, G. and Wijayagunasekara, H.H. (1981). Beobachttngen uber die Anpassung von Varroa jacobsoni an ihren naturlichen wirt Apis cerana in Sri Lanka. Apidologie 12: 37-40. Kuenen, L.P.S. and Calderone, N.W. (2000). Varroa mite infestations in elevated honey bee brood cells: Effect of context and caste. J. Insect Behav. 13: 201-215. Message, D. (1986). Aspectos reprodutivos do ácaro Varroa jacobsoni e seus efeitos em colônias de abelhas africanizadas. M.Sc. thesis, Faculdade de Medicina de Ribeirão Preto, USP, Ribeirão Preto, SP, Brazil. Message, D. and Gonçalves, L.S. (1995). Effect of the size of worker brood cells of Africanized honey bees on infestation and reproduction of the ectoparasitic mite Varroa jacobsoni Oud. Apidologie 26: 381-386. Moretto, G., Gonçalves, L.S. and De Jong, D. (1991). The effects of climate and bee race on Varroa jacobsoni Oud. infestations in Brazil. Apidologie 22: 197-203. Ritter, W. and De Jong, D. (1984). Reproduction of Varroa jacobsoni Oud. in Europe, the middle East and tropical South America. Z. Angew. Entomol. 98: 55-57. |
|